Better Manage Your Risk Assessment
Evaluation of the impact of critical process parameters (CPP) on critical quality attributes (CQA) is required during drug development and when scaling manufacturing or transferring to another site. DeltaV Process Risk Assessment (PRA), part of Emerson’s Process Knowledge Management software suite makes managing the risk assessment activities easier.

Seamless Integration for Easier Risk Assessments
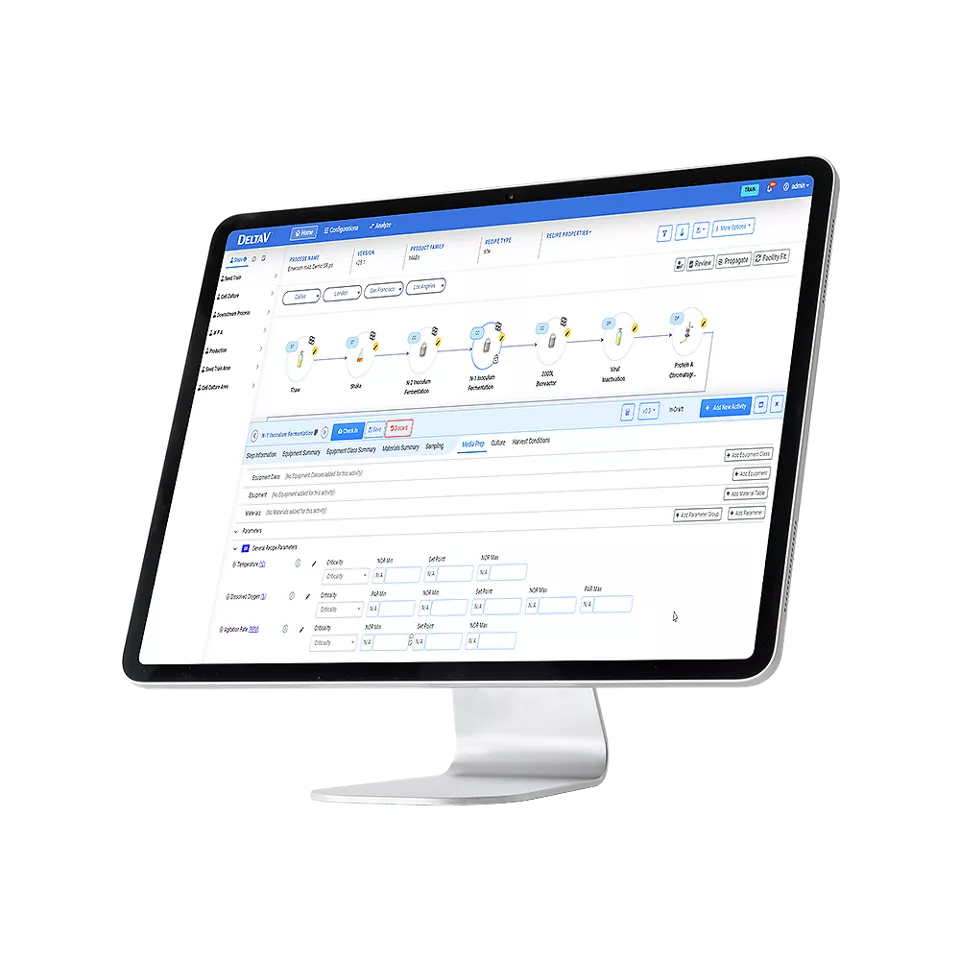
DeltaV PSM allows users to identify CPPs and CQAs within manufacturing specifications. Since DeltaV PRA has full access to existing recipes built in DeltaV PSM, there is no need to manually re-enter critical process information as occurs with other risk assessment solutions, saving considerable time and effort and preventing errors.
Risk Studies
DeltaV PRA enables users to effortlessly create different types of quantitative studies. These include:

Cause and Effect (C&E)
Help correlate CPPs and CQAs by ranking the impact of CPPs on CQAs

Failure Mode Effect Analysis (FMEA)
Identify the potential ways a process might fail and evaluate the impact of each failure mode

Parameter Study
Combine and summarize information across multiple studies in a single table

Functional Relationship Table
Display the relationship between parameters across multiple C&E studies
Key Features
Centralized Management of Critical Parameters
DeltaV PRA provides centralized management of CPPs and CQAs to create a single source of reference for risk assessments.

Explore Complementary Solutions
The DeltaV Process Knowledge Management software suite includes solutions for simplified risk assessment and technology transfer. These leverage the standardized process specifications and seamless integration capabilities provided by DeltaV PSM.




