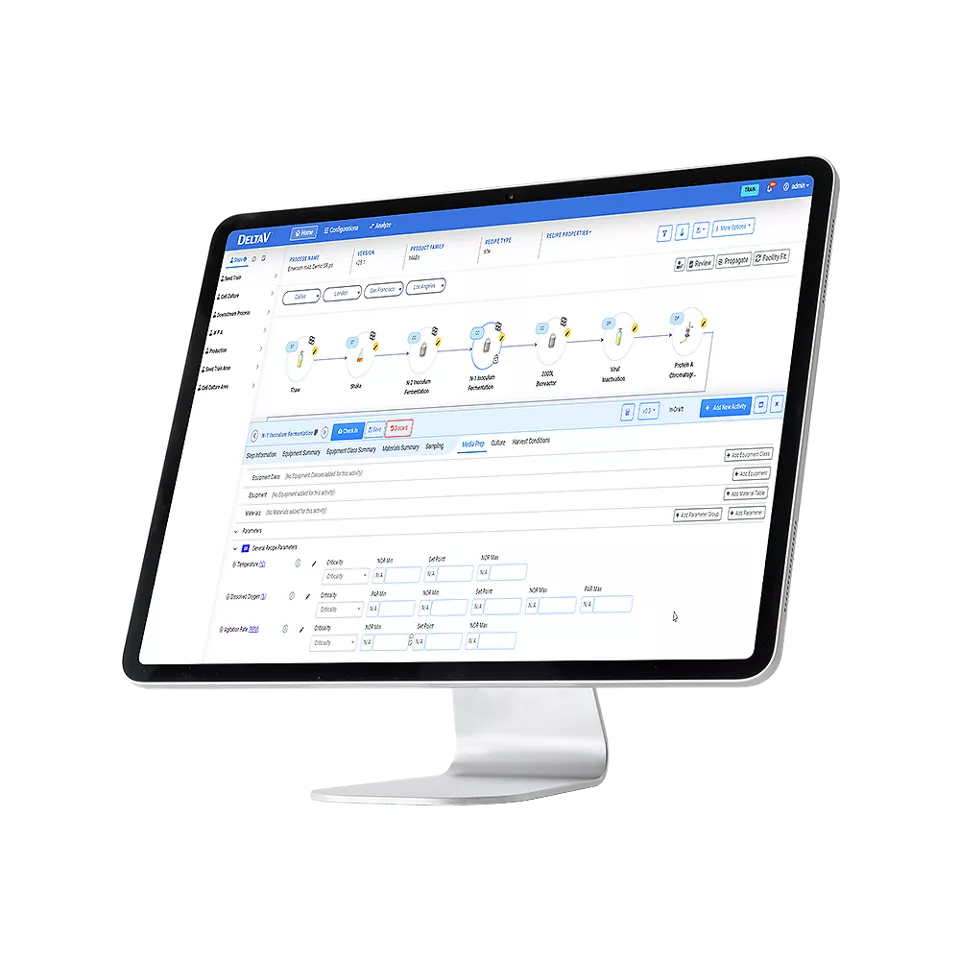
Part of Emerson’s Process Knowledge Management suite of software, DeltaV Recipe Transfer Management (RTM) helps reduce the technology transfer process to almost one click. DeltaV RTM achieves this by enabling seamless integration of data from the most commonly used software applications into DeltaV Process Specification Management (PSM) and the simplified transfer of process parameters to production systems.

Integration of most commonly used software
To enable DeltaV PSM to create a continuum of product and process knowledge throughout the entire drug development lifecycle, DeltaV RTM facilitates integration of other systems, such as enterprise resource planning (ERP), electronic lab notebooks (ELN), and laboratory information management systems (LIMS).

Technology transfer
DeltaV RTM facilitates automatic transfer of process parameters from DeltaV PSM to production systems such as the distributed control system (DCS) and manufacturing execution system (MES). This significantly reduces the time required to both perform technology transfers and manage process parameter changes.

External application interfaces
DeltaV RTM uses an integration framework to create different external application interfaces (EAI), also known as extensions, that are decoupled from DeltaV Process Specification Management (PSM) software. EAIs enable external applications to seamlessly exchange data with DeltaV PSM. DeltaV RTM can create three main types of extensions:
- Application Programming Interface (API) extensions
- Data Pipeline Extensions (DPE)
- Report Extensions
Key Features
Flexible Integration Framework
Multiple interfaces are available for exchanging different data formats to and from DeltaV PSM.

Explore Complementary Solutions
The DeltaV Process Knowledge Management software suite includes solutions for simplified risk assessment and technology transfer. These leverage the standardized process specifications and seamless integration capabilities provided by DeltaV PSM.




