
Streamline and Scale Complex Formulation Processes
Digitally transform formulation operations to improve accuracy, reduce risk, and accelerate time to market
Formulation processes in the biotechnology industry demand a delicate balance of control, precision, and documentation. With integrated automation, batch management, and real-time insights, production teams can improve batch quality, enhance regulatory compliance, and ensure the consistency required for high-value biologics. Scalable solutions support seamless technology transfer from clinical to commercial production, enabling flexibility and reliability throughout the drug development lifecycle.
Formulation Solutions in Action
Whether you're developing complex biologics or scaling up a commercial production line, modern formulation solutions offer end-to-end visibility and control. Real-time batch performance monitoring, recipe management, and digital documentation help reduce variability, improve quality assurance, and support audit readiness. Proven across global biotech facilities, these technologies are tailored to meet evolving regulatory expectations while delivering operational efficiency.
Optimize Formulation, Fill, and Finish Manufacturing Facilities

With Emerson’s solutions, you can efficiently manage materials and equipment to ensure on-time production, integrate planning and control systems, and initiate formulation changes quickly and seamlessly.
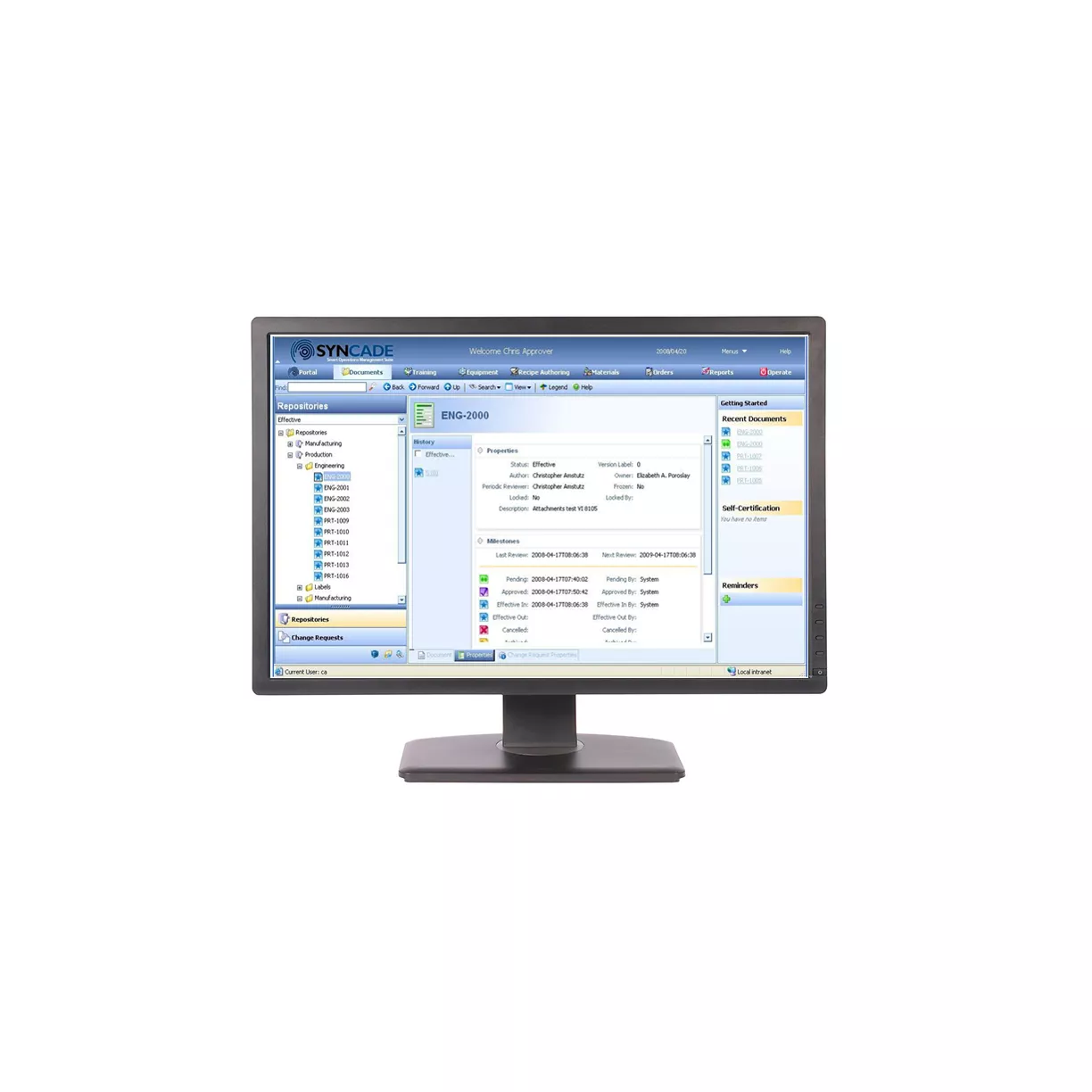
DeltaV™ Manufacturing Execution System
Gain real-time visibility and control over batch execution with integrated data and recipe management.
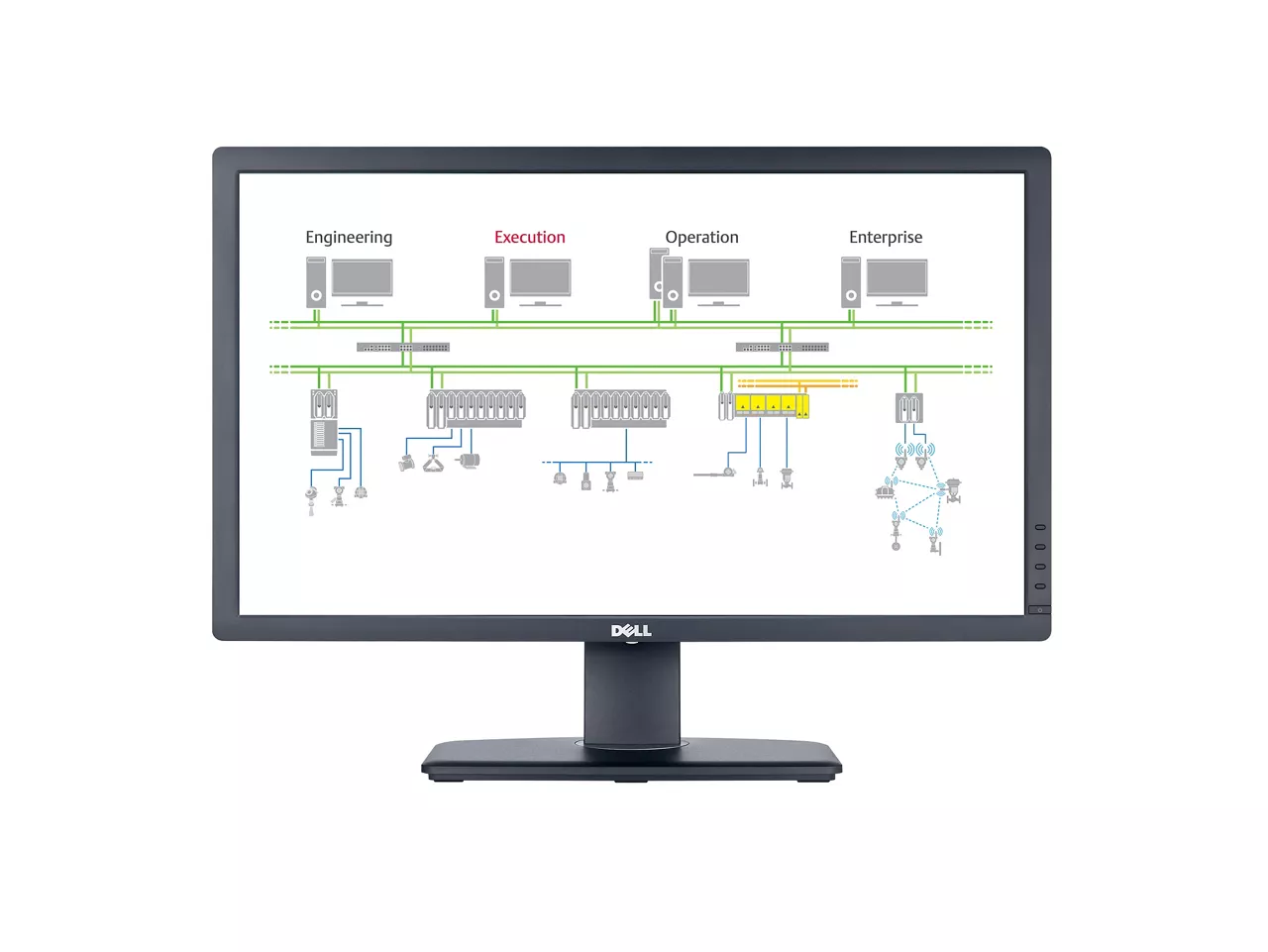
DeltaV™ Batch Executive
Coordinate and automate batch processes with flexible recipe configuration and consistent execution.

AMS™ for Reliability
Asset monitoring helps businesses track and monitor their physical assets. It provides real-time data on the location, condition, and performance of assets, allowing businesses to make informed decisions and optimize their operations.
Solution-Related Documents for Formulation
Access application notes, case studies, and technical documentation that demonstrate how Emerson’s solutions optimize formulation processes. From accurate dosing and homogenous mixing to consistent batch production, our resources highlight strategies to improve efficiency, ensure compliance, and scale with confidence.
Frequently Asked Questions (FAQs)
Explore frequently asked questions to gain a deeper understanding of formulation processes, technologies, and compliance considerations in biotech manufacturing.
Formulation involves combining active pharmaceutical ingredients with excipients to create a stable, effective drug product suitable for storage and administration.
Automation improves batch consistency, reduces manual errors, enhances data integrity, and supports compliance with regulatory requirements.
Challenges include maintaining sterility, managing complex recipes, scaling up from clinical to commercial production, and ensuring batch-to-batch consistency.
Unreliable equipment can lead to process deviations, contamination risks, and downtime, ultimately impacting product quality and regulatory compliance.
Data integrity ensures that records are accurate, complete, and auditable, which is critical for compliance and traceability in regulated environments.
MES and batch systems provide real-time data, enforce recipe parameters, automate reporting, and facilitate electronic records for regulatory compliance.







