
Producing with Certainty and Agility
Smart systems that enable repeatable, compliant, and efficient biomanufacturing
Batch processes have become increasingly complex for life science companies, particularly on the pharmaceutical production side where proper data measurement and analysis is essential to profitability. Emerson offers intelligent automated batch solutions that not only can provide accurate data measurement, but also reduce variability, enhance product utilization, and maximize yield for more effective batch processes.
Simplify and Optimize Batch Processes
Whether producing monoclonal antibodies or vaccines, batch processes demand repeatable performance and real-time insight. Modern platforms integrate scheduling, automation, and data management into a unified environment that supports electronic batch records, analytics, and ongoing improvements. By reducing manual interventions and centralizing operations, teams can improve throughput while maintaining strict adherence to GMP standards.
Technologies That Power End-to-End Batch Control

Enable continuous manufacturing with automation.
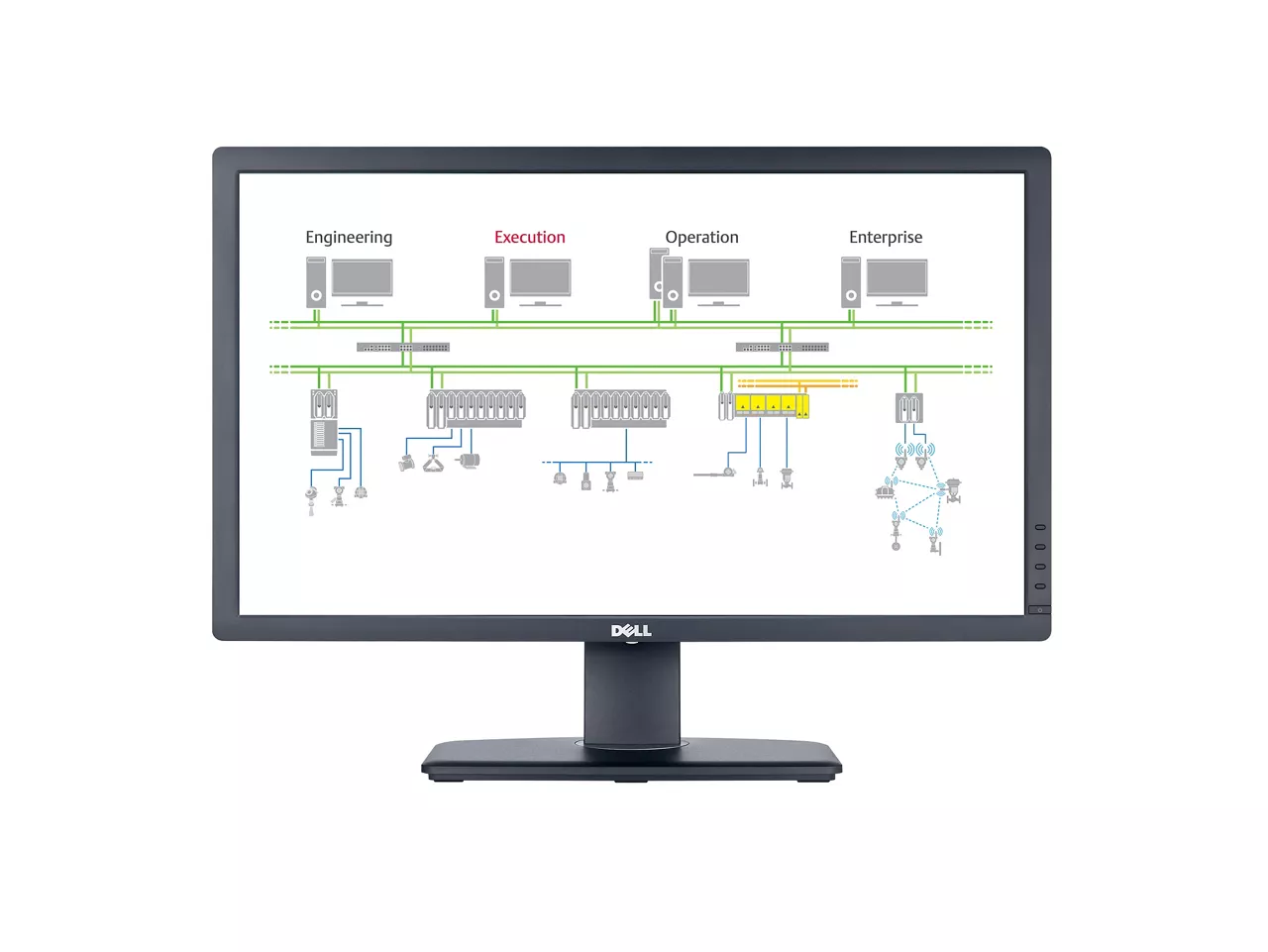
DeltaV™ Batch Executive
Centralized recipe management and execution platform for full batch process control enables repeatability, auditability, and scalability across operations.
DeltaV™ Manufacturing Execution System
Digitally manages electronic batch records, materials, and equipment tracking, and improves visibility and reduces documentation effort.

AMS for Reliability
Predictive maintenance platform that integrates with batch operations to minimize downtime and preserve product quality.
Solutions for High-Yield Manufacturing and Efficient Fluid Transfer

Keeping up with high-yield manufacturing and efficient fluid transfer is critical to the purification and filtration processes. Skids are crucial to prevent damage to products and equipment. Emerson’s fluid control and pneumatics technologies allow you to closely monitor complex processes, prevent damage to the product, and retain high throughput.

DeltaV PK Controller
The DeltaV PK Controller brings faster logic execution, built-in native Ethernet device protocols, and many scalable sizes, enabling it to address a wide variety of applications.

DeltaV™ Batch Executive
The DeltaV Batch Executive manages everything from recipe execution to history collection. It is responsible for carrying out batch procedures, coordinating communication between phases, and allocating equipment and other resources required by a batch.

DeltaV™ Manufacturing Execution System
Unlock higher productivity and streamlined operations in manufacturing with DeltaV’s cutting-edge MES solutions.
Advanced Fill Finish and Packaging Control

Discover how manufacturers are using Emerson’s single-use technologies to streamline fill finish, and packaging operations. These solutions support high-yield production, maintain sterility, and enable fast changeovers—helping you reduce contamination risk, improve throughput, and meet regulatory requirements with confidence.

DeltaV PK Controller
The DeltaV PK Controller brings faster logic execution, built-in native Ethernet device protocols, and many scalable sizes, enabling it to address a wide variety of applications.

DeltaV™ Batch Executive
The DeltaV Batch Executive manages everything from recipe execution to history collection. It is responsible for carrying out batch procedures, coordinating communication between phases, and allocating equipment and other resources required by a batch.

DeltaV™ Manufacturing Execution System
Unlock higher productivity and streamlined operations in manufacturing with DeltaV’s cutting-edge MES solutions.
Solution-Related Documents for Batch
Access a curated library of technical documents, case studies, and solution overviews designed to support batch process optimization. These resources demonstrate how Emerson’s technologies help enhance repeatability, streamline regulatory compliance, and drive greater efficiency across every stage of batch production.
Business Groups in Batch
Successful batch manufacturing requires more than a single system—it needs synchronized technologies that span control, measurement, and asset reliability. Business groups across the portfolio offer specialized tools that enable consistent batch outcomes, reduce variability, and improve compliance. Together they create an environment where data, processes, and people are connected for long-term Operational Excellence.
Automation Systems
Frequently Asked Questions (FAQs)
Explore answers to frequently asked questions about batch processing in biotechnology manufacturing. Learn how automation, recipe management, and digital batch records support consistent quality, regulatory compliance, and efficient scale-up from clinical to commercial production.
Batch processing is a production method where a specific quantity of a drug product is produced in a single, defined run. It is commonly used in biotechnology to ensure consistency, traceability, and regulatory compliance across multiple stages of drug development and manufacturing.
Batch automation streamlines execution by minimizing manual interventions, ensuring recipe adherence, and providing real-time monitoring and control. This leads to faster production cycles, reduced errors, and enhanced product quality.
Core components include a batch execution engine, recipe management, operator interface, real-time data capture, electronic records, and integration with MES and automation systems.
ISA-88 is a widely adopted standard for batch process control. It provides a framework for defining recipes, equipment modules, and control strategies, supporting scalable and flexible batch operations in biotechnology manufacturing.
Batch processing is discrete and time-bound, which is ideal for high-mix, low-volume production. Continuous processing runs non-stop and is used for high-volume, consistent product manufacturing. Batch remains more common in biologics due to complexity and regulatory factors.
Effective recipe management ensures precise ingredient quantities, consistent processing steps, and repeatable results, which are critical in the production of biologics and other high-value therapeutics.







